Evaluation of Serum Copper, Zinc, Iron, and Calcium Levels among Children with Autism Spectrum Disorder and Attention-Deficit/ Hyperactivity Disorder in Baghdad
DOI:
https://doi.org/10.32007/jfacmedbaghdad3225Keywords:
Attention-Deficit/Hyperactivity Disorder, Autism Spectrum Disorder, Heavy Metals, Neurodevelopmental disorders, Trace elementsAbstract
Background: Autism spectrum disorder (ASD) and attention-deficit/hyperactivity disorder (ADHD) are neurodevelopmental conditions that arise from a complex interplay of genetic, biological, and environmental factors. Essential metal elements (EMEs), including copper, zinc, iron, and calcium, support brain development, support neuronal communication, and maintain normal metabolic activity. Any increase or decrease in these elements may play a role in the development of ASD and ADHD.
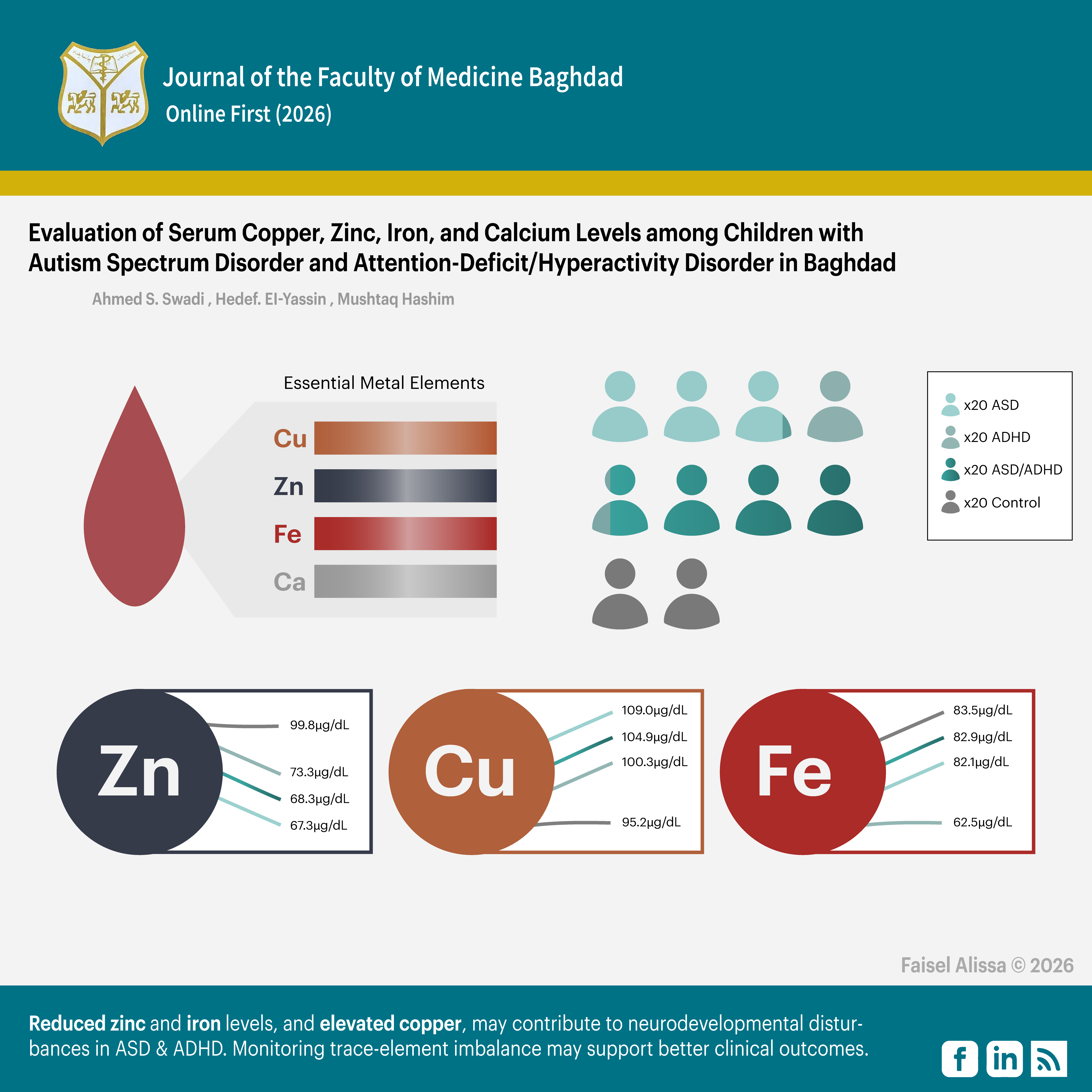
Objectives: The study aimed to investigate the relationship between serum levels of zinc, copper, iron, and calcium with ASD and ADHD. Moreover, to compare these levels among children with ASD, ADHD, combined ASD plus ADHD, and healthy controls.
Methods: A case-control study was carried out at the College of Medicine, University of Baghdad, between March and September 2025. In total, 200 children aged 2–15 years were included and divided into four groups: Controls (n = 40), ADHD (n = 30), ASD (n = 57), and ASD + ADHD (n = 73). Five ml of venous blood from each child was dispensed into a gel tube to estimate zinc, copper, iron, and calcium. Assessment of inorganic elements zinc and copper was performed by Flam atomic absorption spectrometry (FAAS) while iron and calcium was performed by spectrophotometer.
Results: Serum zinc levels were significantly lower in all patient groups compared with the controls. Copper levels were significantly higher, particularly in the ADHD and ASD + ADHD groups. Serum calcium showed no significant differences among the groups. Serum iron levels were significantly reduced only in ADHD group.
Conclusion: Reduced zinc and iron levels, together with elevated copper, may contribute to neurodevelopmental disturbances in ASD and ADHD. Monitoring and correcting trace-element imbalance could support better clinical outcomes in affected children.
Received: Nov. 2025
Revised: Jan. 2026
Accepted: Jan. 2026
Published online: Mar. 2026
Published: April 2026
References
1.Dou JF, Schmidt RJ, Volk HE, et al. Exposure to heavy metals in utero and autism spectrum disorder at age 3: a meta-analysis of two longitudinal cohorts of siblings of children with autism. Environ Health. 2024;23(1):62. https://doi.org/10.1186/s12940-024-01101-2. DOI: https://doi.org/10.1186/s12940-024-01101-2
2. Sauer AK, Stanton JE, Hans S, Grabrucker AM. Autism Spectrum Disorders: Etiology and Pathology. In: Grabrucker AM, editor. Autism Spectrum Disorders. Brisbane (AU): Exon Publications; 2021. 1-5. https://doi.org/10.36255/exonpublications.autismspectrumdisorders.2021.etiology. DOI: https://doi.org/10.36255/exonpublications.autismspectrumdisorders.2021.etiology
3. Tang S, Zhang G, Ran Q, et al. Quantitative susceptibility mapping shows lower brain iron content in children with attention-deficit hyperactivity disorder. Hum Brain Mapp. 2022;43(8):2495-502. https://doi.org/10.1002/hbm.25798. DOI: https://doi.org/10.1002/hbm.25798
4. Hussein RA, Refai RH, El-Zoka AH, et al. Association between some environmental risk factors and attention-deficit hyperactivity disorder among children in Egypt: a case-control study. Ital J Pediatr. 2025;51(1):19. https://doi.org/10.1186/s13052-025-01843-w. DOI: https://doi.org/10.1186/s13052-025-01843-w
5. Zheng Y, Pingault JB, Unger JB, et al. Genetic and environmental influences on attention-deficit/hyperactivity disorder symptoms in Chinese adolescents: a longitudinal twin study. Eur Child Adolesc Psychiatry. 2020;29(2):205-16. https://doi.org/10.1007/s00787-019-01346-0. DOI: https://doi.org/10.1007/s00787-019-01346-0
6. Kim JH, Kim JY, Lee J, et al. Environmental risk factors, protective factors, and peripheral biomarkers for ADHD: an umbrella review. Lancet Psychiatry.2020;7(11):955-70. https://doi.org/10.1016/S2215-0366(20)30312-6. DOI: https://doi.org/10.1016/S2215-0366(20)30312-6
7. Zhang P, Georgiou CA, Brusic V. Elemental metabolomics. Brief Bioinform. 2018;19(3):524-36. https://doi.org/10.1093/bib/bbw131. DOI: https://doi.org/10.1093/bib/bbw131
8. Zoroddu MA, Aaseth J, Crisponi G, et al. The essential metals for humans: a brief overview. J Inorg Biochem.2019;195:120-9. https://doi.org/10.1016/j.jinorgbio.2019.03.013. DOI: https://doi.org/10.1016/j.jinorgbio.2019.03.013
9. Ma J, Wu J, Li H, et al. Association Between Essential Metal Elements and the Risk of Autism in Chinese Han Population. Biol Trace Elem Res. 2022;200(2):505-15. https://doi.org/10.1007/s12011-021-02690-6. DOI: https://doi.org/10.1007/s12011-021-02690-6
10. Al-Naama N, Mackeh R, Kino T. C2H2-Type Zinc Finger Proteins in Brain Development, Neurodevelopmental, and Other Neuropsychiatric Disorders: Systematic Literature-Based Analysis. Front Neurol. 2020;11:32. https://doi.org/10.3389/fneur.2020.00032. DOI: https://doi.org/10.3389/fneur.2020.00032
11. Bourassa D, Elitt CM, McCallum AM, et al. Chromis-1, a Ratiometric Fluorescent Probe Optimized for Two-Photon Microscopy Reveals Dynamic Changes in Labile Zn(II) in Differentiating Oligodendrocytes. ACS Sens. 2018;3(2):458-67. https://doi.org/10.1021/acssensors.7b00887. DOI: https://doi.org/10.1021/acssensors.7b00887
12. da Silva PR, do Nascimento Gonzaga TK, Maia RE, et al . Ionic channels as potential targets for the treatment of autism spectrum disorder: a review. CurrNeuropharmacol.2022;20(10):1834-49. https://doi.org/10.2174/1570159X19666210809102547. DOI: https://doi.org/10.2174/1570159X19666210809102547
13. Długosz A, Wróblewski M, Błaszak B, et al. The Role of Nutrition, Oxidative Stress, and Trace Elements in the Pathophysiology of Autism Spectrum Disorders. Int J Mol Sci. 2025;26(2):808. https://doi.org/10.3390/ijms26020808. DOI: https://doi.org/10.3390/ijms26020808
14. Telianidis J, Hung YH, Materia S, et al. Role of the P-Type ATPases, ATP7A and ATP7B in brain copper homeostasis. Front Aging Neurosci. 2013;5:44. https://doi.org/10.3389/fnagi.2013.00044. DOI: https://doi.org/10.3389/fnagi.2013.00044
15. Li SO, Wang JL, Bjørklund G, et al. Serum copper and zinc levels in individuals with autism spectrum disorders. Neuroreport. 2014;25(15):1216-20. https://doi.org/10.1097/WNR.0000000000000251. DOI: https://doi.org/10.1097/WNR.0000000000000251
16. Robberecht H, Verlaet AAJ, Breynaert A, et al. Magnesium, iron, zinc, copper and selenium status in attention-deficit/hyperactivity disorder (ADHD). Molecules.2020;25(19):4440. https://doi.org/10.3390/molecules25194440. DOI: https://doi.org/10.3390/molecules25194440
17. Nanou E, Catterall WA. Calcium channels, synaptic plasticity, and neuropsychiatric disease. Neuron.2018;98(3):466-81. https://doi.org/10.1016/j.neuron.2018.03.017. DOI: https://doi.org/10.1016/j.neuron.2018.03.017
18. do Nascimento PK, Oliveira Silva DF, de Morais TL, et al. Zinc status and Autism Spectrum Disorder in children and Adolescents: A Systematic Review. Nutrients.2023;15(16):3663. https://doi.org/10.3390/nu15163663. DOI: https://doi.org/10.3390/nu15163663
19. Prasad AS. Zinc in Human Health: Effect of Zinc on Immune Cells. Mol Med. 2008; 14(5-6): 353-357.
https://doi.org/10.2119/2008-00033.Prasad. DOI: https://doi.org/10.2119/2008-00033.Prasad
20. Ahmadani A, Kittana M, Al-Marzooq F, et al. Zinc ion dyshomeostasis in autism spectrum disorder. Nutr Res Rev. 2025;38(2):661-681. https://doi.org/10.1017/S095442242500006X. DOI: https://doi.org/10.1017/S095442242500006X
21. Chen X, Jiang Y, Wang Z, et al. Alteration in gut microbiota associated with zinc deficiency in school-age children. Nutrients. 2022;14(14):2895. https://doi.org/10.3390/nu14142895. DOI: https://doi.org/10.3390/nu14142895
22. Gibson RS, Bailey KB, Ferguson EL. A review of phytate, iron, zinc, and calcium concentrations in plant-based complementary foods used in low-income countries and implications for bioavailability. Food Nutr Bull. 2010; 31(2 Suppl2): S134-46. https://doi.org/10.1177/15648265100312S206. DOI: https://doi.org/10.1177/15648265100312S206
23. El-Saadany NZH, Abdel-Fattah MF, Siam AG, et al. Relationship between Serum Iron Level and Zinc Level with Attention Deficit Hyperactivity Disorder in Children. Egypt J Hosp Med. 2022;87(1):2106-12. https://doi.org/10.21608/ejhm.2022.232828. DOI: https://doi.org/10.21608/ejhm.2022.232828
24. Yang R, Zhang Y, Gao W, et al. Blood levels of trace elements in children with attention-deficit hyperactivity disorder: results from a case-control study. Biol Trace Elem Res. 2019;187(2):376-82. https://doi.org/10.1007/s12011-018-1408-9. DOI: https://doi.org/10.1007/s12011-018-1408-9
25. Siddiqi UR, Begum S, Shahjadi S, et al. Plasma zinc, copper and serum ceruloplasmin levels of autism spectrum disorder children in Bangladesh. Heliyon.2023;9(8):e18624. https://doi.org/10.1016/j.heliyon.2023.e18624. DOI: https://doi.org/10.1016/j.heliyon.2023.e18624
26. Zhang XH, Yang T, Chen J, et al. Association between Serum Trace Elements and Core Symptoms in Children with Autism Spectrum Disorder: A National Multicenter Survey. Chinese Journal of Contemporary Pediatrics. 2021;23(5):445-50. https://doi.org/10.7499/j.issn.1008-8830.2101163 .
27. Escobedo-Monge MF, Barrado E, Parodi-Roman J, et al. Copper/Zinc ratio in Childhood and Adolescence: A Review. Metabolites. 2023;13(1):82. https://doi.org/10.3390/metabo13010082. DOI: https://doi.org/10.3390/metabo13010082
28. Linder MC. Biochemistry of copper. New York: Springer Science+Business Media; 2013. https://books.google.com/books/about/Biochemistry_of_Copper.html?id=8x4DCAAAQBAJ&utm_source.
29. Nayak S, Sahu S, Patra S, et al. Assessment of Copper and Zinc Levels in Hair and Urine of Children with Attention Deficit Hyperactivity Disorder: A Case-Control Study in Eastern India. Cureus. 2021;13(12):e20692. https://doi.org/10.7759/cureus.20692. DOI: https://doi.org/10.7759/cureus.20692
30. Rashaid AHB, Nusair SD, Alqhazo MT, et al. Heavy metals and trace elements in scalp hair samples of children with severe Autism spectrum disorder: A case-control study on Jordanian children. J Trace Elem Med Biol. 2021;67:126790. https://doi.org/10.1016/j.jtemb.2021.126790. DOI: https://doi.org/10.1016/j.jtemb.2021.126790
31. Beard JL. Iron Biology in Immune Function, Muscle Metabolism and Neuronal Function. J Nutr. 2001; 131(2): 568S-580S. https://doi.org/10.1093/jn/131.2.568S. DOI: https://doi.org/10.1093/jn/131.2.568S
32. Ganz T. Hepcidin and iron regulation, 10 years later. Blood. 2011; 117(17): 4425-4433. https://doi.org/10.1182/blood-2011-01-258467. DOI: https://doi.org/10.1182/blood-2011-01-258467
33. Konofal E, Lecendreux M, Deron J, et al. Effects of iron supplementation on attention deficit hyperactivity disorder in children. Pediatr Neurol. 2008;38(1):20-6. https://doi.org/10.1016/j.pediatrneurol.2007.08.014. DOI: https://doi.org/10.1016/j.pediatrneurol.2007.08.014
34. Skalny AV, Simashkova NV, Klyushnik TP, et al. Assessment of serum trace elements and electrolytes in children with childhood and atypical autism. J Trace Elem Med Biol. 2017;43:9-14. https://doi.org/10.1016/j.jtemb.2016.09.009. DOI: https://doi.org/10.1016/j.jtemb.2016.09.009
35. Yu E, Sharma S. Physiology, calcium [Updated 2023 Aug 14]. StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2026 jan. https://www.ncbi.nlm.nih.gov/books/NBK482128/ (accessed 25 March 2026).
36. Klocke B, Krone K, Tornes J, et al. Insights into the role of Intracellular Ca2+ signaling in neurodevelopmental disorders. Front Neurosci. 2023;17:1093099. https://doi.org/10.3389/fnins.2023.1093099. DOI: https://doi.org/10.3389/fnins.2023.1093099
37. Alkhalidy H, Abushaikha A, Alnaser K, et al. Nutritional status of pre-school children and determinant factors of autism: A case-control study. Front Nutr. 2021;8:627011. https://doi.org/10.3389/fnut.2021.627011. DOI: https://doi.org/10.3389/fnut.2021.627011
38. Alzghoul L, Al-Eitan LN, Aladawi M, et al. The association between serum Vitamin D3 levels and autism among Jordanian boys. J Autism Dev Disord. 2020;50(9):3149-54. https://doi.org/10.1007/s10803-019-04017-w. DOI: https://doi.org/10.1007/s10803-019-04017-w
Downloads
Published
Issue
Section
Categories
License
Copyright (c) 2026 Ahmed S. Swadi, Hedef. EI-Yassin, Mushtaq T. Hashim

This work is licensed under a Creative Commons Attribution 4.0 International License.











 Creative Commons Attribution 4.0 International license..
Creative Commons Attribution 4.0 International license..


