Programmed Death Ligand 1 Receptor Protein Expression in a Group of Triple-Negative Breast Carcinoma Patients and its Correlation with the Clinicopathological Parameters
DOI:
https://doi.org/10.32007/jfacmedbaghdad3244Keywords:
Breast carcinoma, Immunotherapies, Immune checkpoint inhibitors, PD-L1 protein, Triple negative breast carcinomaAbstract
Background: Triple-negative breast carcinoma has an aggressive nature, poor prognosis, with high recurrence rates and metastasis. It has a poor response to targeted therapies, leaving a restricted number of efficient treatments, including chemotherapy and radiotherapy. Some types of triple-negative breast carcinoma are considered immunogenic types, which mean that these tumors may be susceptible targets to a new line of treatment known as immunotherapy. Several biomarkers have been discovered to determine patients who could be eligible candidates to receive immunotherapies. One of these biomarkers is the programmed death receptor 1 (PD1)/ programmed death ligand 1 (PD-L1) overexpression.
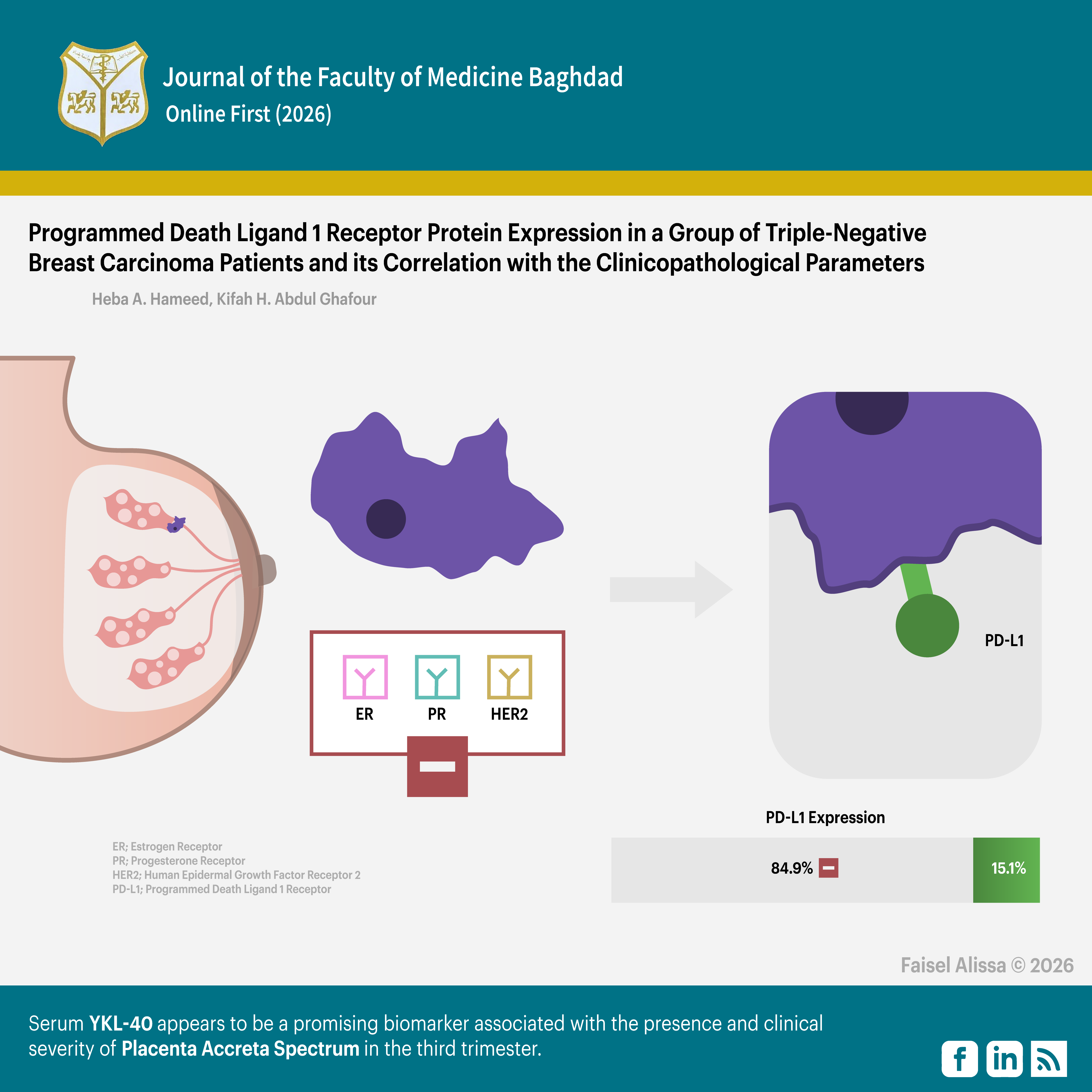
Objectives: To assess the expression status of the PD-L1 protein in a group of triple-negative breast carcinoma patients, and its correlation with the clinicopathological parameters.
Methods: The study was conducted from October 2024 to April 2025. A cross-sectional study involved 53 patients, who were diagnosed as triple-negative invasive ductal breast carcinoma. The formalin-fixed paraffin-embedded blocks were retrieved from the archives department of the histopathological laboratories in the Medical City Complex hospitals, Baghdad, Iraq. The clinical data of these patients were reviewed, and the blocks were sectioned, prepared, and stained with monoclonal anti-PD-L1 antibody.
Results: The majority of the cases (45 patients, 84.9%) had negative expression of PD-L1 protein, while eight patients (15.1%) had positive expression of PD-L1 protein. A non-significant correlation was found between the PD-L1 protein expression and the clinicopathological parameters.
Conclusion: The vast majority of the studied cases had a negative expression of PD-L1 protein. It seemed that the correlation between PD-L1 expression status and the studied clinicopathological parameters was not significant.
Received: Dec. 2025
Revised: April 2026
Accepted: March 2026
Published Online: March 2026
Published: April 2026
References
1. Collins L.C. Breast. In: Goldblum JR, Lamps LW, McKenny JK, editors. Rosai and Ackerman’s surgical pathology. 11th edition. Philadelphia: Elsevier Inc.; 2018. p. 1434-1527. https://elsevier.com/books/rosai-and-ackermans-surgical-pathology-2-volume-set/goldblum/978-0-323-26339-9.
2. World Health Organization. Breast cancer. https://www.who.int/news-room/fact-sheets/detail/breast-cancer.
3. Iraqi Cancer Registry 2023. Iraqi Cancer Board. Ministry of Health, Republic of Iraq, 2023. https://storage.moh.gov.iq/2024/11/24/2024_11_24_12127028949_4299728097670824.pdf
4. Tan PH, Ellis I, Allison K, et al. World Health Organization classification of tumors – breast tumors, 5th edition Vol. 2, Lyon (France), IARC, 2019 https://publications.iarc.fr/581
5. Zhu Y, Zhu X, Tang C, et al. Progress and challenges of immunotherapy in triple-negative breast cancer. BBA Reviews on Cancer. 2021 Dec 1;1876(2): 188593. https://doi:org/10.1016/j.bbcan.2021.188593. DOI: https://doi.org/10.1016/j.bbcan.2021.188593
6. Stanowska O, Kuczkiewicz-Siemion O, Dębowska M, et al. PD-L1 positive high-grade triple-negative breast cancer patients respond better to standard neoadjuvant treatment- a retrospective study of PD-L1 expression in relation to different clinicopathological parameters. J. Clin. Med. 2022 Sep 21;11(19): 5524. https://doi.org/10.3390/jcm11195524. DOI: https://doi.org/10.3390/jcm11195524
7. Chen L, Zhou H, Wu H, et al. Effect of immunotherapy or anti-angiogenesis therapy combined with chemotherapy for advanced triple-negative breast cancer: A real-world retrospective study. Int Immunopharmacol. 2024 Dec 25; 143:113516. https://doi.org/10.1016/j.intimp.2024.113516. DOI: https://doi.org/10.1016/j.intimp.2024.113516
8. Kossai M, Radosevic-Robin N, Penault-Llorca F. Refining patient selection for breast cancer immunotherapy: beyond PD-L1. ESMO Open. 2021 Oct 1;6(5):100257. https://doi.org/10.1016/j.esmoop.2021.100257. DOI: https://doi.org/10.1016/j.esmoop.2021.100257
9. Keenan TE, Tolaney SM. Role of immunotherapy in triple-negative breast cancer. J Natl Compr Canc Netw. 2020 Apr 1;18(4): 479-89. https://doi.org/10.6004/jnccn.2020.7554. DOI: https://doi.org/10.6004/jnccn.2020.7554
10. Akhtar M, Rashid S, Al-Bozom IA. PD-L1 immunostaining: what pathologists need to know. Diagn Pathol. 2021 Oct 25; 16(1):94. https://doi.org/10.1186/s13000-021-01151-x. DOI: https://doi.org/10.1186/s13000-021-01151-x
11. Sukumar J, Gast K, Quiroga D, et al. Triple-negative breast cancer: promising prognostic biomarkers currently in development. Expert Rev Anticancer Ther. 2021 Feb 1;21(2): 135-48. https://doi.org/10.1080/14737140.2021.1840984. DOI: https://doi.org/10.1080/14737140.2021.1840984
12. Li CJ, Lin LT, Hou MF, et al. PD-L1/PD1 blockade in breast cancer: the immunotherapy era (Review). Oncol Rep. 2021 Jan 1;45(1): 5-12. https://doi.org/10.3892/or.2020.7831. DOI: https://doi.org/10.3892/or.2020.7831
13. Porta FM, Sajjadi E, Venetis K, et al. Immune biomarkers in triple-negative breast cancer: improving the predictivity of current testing methods. J Pers Med. 2023 Jul 23;13(7):1176. https://doi.org/10.3390/jpm13071176. DOI: https://doi.org/10.3390/jpm13071176
14. Agilent Technologies. PD-L1 IHC 22C3 pharmDx interpretation Manual-Triple Negative Breast Cancer. https://www.agilent.com/cs/library/usermanuals/public/29389_22c3_pharmdx_tnbc_interpretation_manual_kn355.pdf.
15. Troschel FM, Minte A, Ismail YM, et al. Knockdown of Musashi RNA binding protein decreases radioresistance but enhances cell motility and invasion in triple-negative breast cancer. Int J Mol Sci. 2020 Mar 21; 21(6): 2169. https://doi.org/10.3390/ijms21062169. DOI: https://doi.org/10.3390/ijms21062169
16. Schmidt G, Guhl MM, Solomayer EF, et al. Immunohistochemical assessment of PD-L1 expression using three different monoclonal antibodies in triple negative breast cancer patients. Arch Gynecol Obstet. 2022 Nov;306(5): 1689-95. https://doi.org/10.1007/s00404-022-06529-w. DOI: https://doi.org/10.1007/s00404-022-06529-w
17. Lee SE, Park HY, Lim SD, et al. Concordance of programmed death-ligand 1 expression between SP142 and 22C3/SP263 assays in triple-negative breast cancer. J Breast Cancer. 2020 Jun 1;23(3): 303. https://doi.org/10.4048/jbc.2020.23.e37. DOI: https://doi.org/10.4048/jbc.2020.23.e37
18. Rammal R, Goel K, Motanagh SA, et al. Immunohistochemical profile of triple-negative breast cancer: SOX10 and AR dual negative tumors have worse outcome. Mod Pathol. 2024 Jul 1; 37(7): 100517. https://doi.org/10.1016/j.modpat.2024.100517. DOI: https://doi.org/10.1016/j.modpat.2024.100517
19. Hammouda SB, Abdessayed N, Abdeljelil NB, et al. Triple negative breast cancer: clinical, pathological and molecular characteristics. Pan Afr Med J. 2022 May 12; 42:30. https://doi.org/10.11604/pamj.2022.42.30.28464.
20. Hu H, Tong K, Tsang JY, et al. Subtyping of triple-negative breast cancers: its prognostication and implications in diagnosis of breast origin. ESMO Open. 2024 Apr 1;9(4):102993. https://doi.org/10.1016/j.esmoop.2024.102993. DOI: https://doi.org/10.1016/j.esmoop.2024.102993
21. Hermansyah D, Kurniawan D, Lesmana DK, et al. The PD-L1 expression among triple-negative breast cancer patients in Universities Sumatera Utara Teaching Hospital, Indonesia. Acta Inform Med. 2022 Dec;30(4):308. https://doi.org/10.5455/aim.2022.30.308-311. DOI: https://doi.org/10.5455/aim.2022.30.308-311
22. Özcan D, Lade-Keller J, Tramm T. Can evaluation of mismatch repair defect and TILs increase the number of triple-negative breast cancer patients eligible for immunotherapy? Pathol Res Pract. 2021 Oct 1;226: 153606. https://doi.org/10.1016/j.prp.2021.153606. DOI: https://doi.org/10.1016/j.prp.2021.153606
23. Ren XY, Song Y, Wang J, et al. Mismatch repair deficiency and microsatellite instability in triple-negative breast cancer: a retrospective study of 440 patients. Front Oncol. 2021 Mar 4;11: 570623. https://doi.org/10.3389/fonc.2021.570623. DOI: https://doi.org/10.3389/fonc.2021.735476
24. Li CY, Zhang S, Zhang XB, et al. Clinicopathological and prognostic characteristics of triple-negative breast cancer (TNBC) in Chinese patients: a retrospective study. Asian Pac J Cancer Prev. 2013;14(6): 3779-84. https://doi.org/10.7314/APJCP.2013.14.6.3779. DOI: https://doi.org/10.7314/APJCP.2013.14.6.3779
25. Horimoto Y, Hlaing MT, Saeki H, et al. Microsatellite instability and mismatch repair protein expression in lymphocyte-predominant breast cancer. Cancer Sci. 2020 Jul; 111(7): 2647-54. https://doi.org/10.1111/cas.14500. DOI: https://doi.org/10.1111/cas.14500
26. Ilieva N, Pencheva M, Hadzhiev H, et al. Impact of neoadjuvant therapy on PD-L1 expression in triple-negative breast cancer and correlation with clinicopathological factors. Diagnostics. 2024 Nov 27;14(23):2672. https://doi.org/10.3390/diagnostics14232672. DOI: https://doi.org/10.3390/diagnostics14232672
27. Mendivelso-González D, Correa PL, Prada-Aceros Net al. Abstract C149: Relationship between PD-L1 expression and clinicopathological characteristics in Hispanic/Latino women with breast cancer. Cancer Epidemiol Biomarkers Prev. 2024 Sep 21;33(9_Supplement):C149-. https://doi.org/10.1158/1538-7755.DISP24-C149. DOI: https://doi.org/10.1158/1538-7755.DISP24-C149
Downloads
Published
Issue
Section
Categories
License
Copyright (c) 2026 Heba A. Al-Saffar, Kifah H. Abdul Ghafour

This work is licensed under a Creative Commons Attribution 4.0 International License.











 Creative Commons Attribution 4.0 International license..
Creative Commons Attribution 4.0 International license..


